Roche Holding Ltd. (ADR)(OTCMKTS:RHHBY) reported promising data from three Phase II trials over two indications for its lead experimental cancer immunotherapy drug atezolizumab. The positive results are going to fetch the attention of industry experts and other market participants in the coming period.
Cancer immunotherapy
It is evident that one aspect of cancer research that is becoming the center of attention is cancer immunotherapy. This avenue comprises of the drugs that are formulated to improve the patient’s immune system to effectively combat cancer. The typical process involves either eliminating or reducing the immunosuppressant quality that permits cancer cells to go unnoticed, supercharging the patient’s immune system to obliterate cancer cells, or a combination of both.
Roche Holding has been the leading force in cancer care with extensive product portfolio and over six dozen underway clinical-stage studies just for oncology. However, the firm I need to come at par with its peers in segment of immunotherapies. With the promising data released on Sunday, the company has taken the first step towards achieving success in the respective segment.
The study
In its first mid-stage trial, named as IMvigor 210, which was highlighted at the European Cancer Congress 2015, checkpoint inhibitor ‘Atezolizumab’, an anti-PDL1 treatment, was evaluated as a therapy for advanced bladder cancer. It should be noted that Advanced bladder cancer is predominantly difficult to treat, but ‘Atezolizumab’ showed a significant 27% objective response rate in mUC patients who had moderate-to-high concentration of PD-L1 expression.
Additionally, a median duration of objective response had not been achieved when Roche announced its results. However, the company stated that 92% of patients with an objective response were responding to the treatment at the time of its data release. The data reported is remarkable as advanced bladder cancer has not recorded a new treatment hit the market in over two decades.
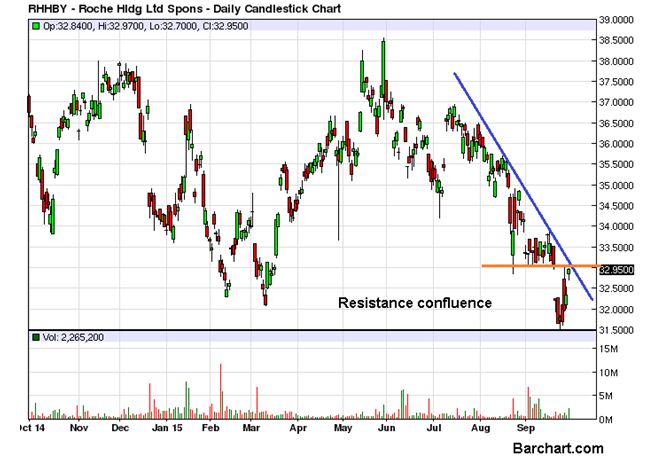
Roche Holding Ltd. (ADR) (OTCMKTS:RHHBY) enjoyed the second consecutive positive day as it finished the last trading session with a gain of 1.92%, that also helped it completely wipe out the big loss incurred on Monday. The volume barely saw a rise as the volume of the day reached 2.2 million against the daily average of 1.9 million but that would be quite natural in a corrective stage. The stock is now facing a very strong resistance confluence zone, consisting of the short term supply area around $33 and the downtrend line, as seen on the chart attached.