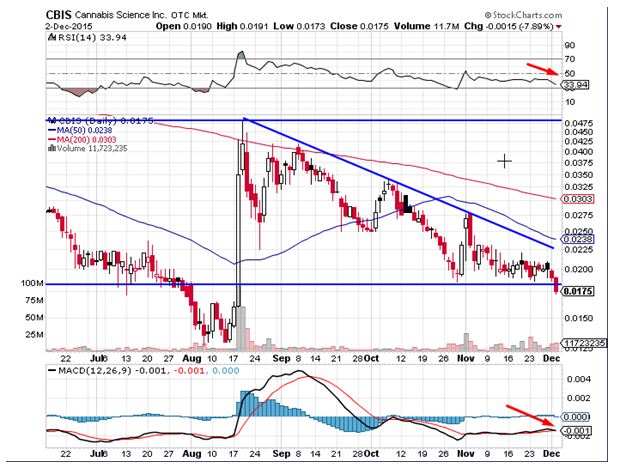
Cannabis Science Inc (OTCMKTS:CBIS) was notable decliners during Wednesday’s trading session declining by 8% on heavy volumes, which were 1.6 times the daily average. The stock has been underperforming the broader markets over the last several months. Cannabis Science has been forming lower tops and lower bottoms indicative of the strong bearish momentum and has broken below the key support level at $0.0187.The oscillators, which measure momentum are clearly indicating that bears are in total control at the current moment. The index for relative strength has given a fresh sell signal, which is considered to be a bearish sign.
Cannabis Science Inc (OTCMKTS:CBIS) announced that it had decided to sign a collaborative drug development deal with IGXBio (GenePro). As per the reports, GenePro is an immunotherapeutic based on DNA structure, which received FDA IND approval recently, pursuant to which it could be used in human trials.
Insights of The Matter
As part of this collaborative drug development deal, these companies will look forward to developing new protocols that could help them explore the synergistic effects of different drugs. Apart from this, they will also work towards the development of new drug candidates in the future.
When asked from Mr. Raymond C. Dabney, CEO & President, Cannabis Science, about this deal, he said that he had a word with IGXBio President Mr. Jim Laufenberg, and CBIS Chief Medical Officer Dr. Allen Herman. Post discussion they all arrived at a conclusion that it was the time for them to accelerate the drug development program.
As part of this collaborative deal, Laufenberg and Herman would come together to select the scientists’ team to successfully carry out research, delineate the shared scope of work between IGXBio and CBIS, and clarify laboratory requirements. Dabney further added that he was working closely with Laufenberg to give this collaboration a final look and ensure that everything fall in line in the best way possible.
IGXBio’s GenePro has already received the approval of FDA, which means that the companies don’t have to waste any time in seeking for approval from FDA in the future. GenePro works effectively on subjects affected by HIV. If developed in the right way, it can resolve one of the biggest and most dangerous health issues haunting human race in today’s time.
Both the firms will carry out different trials in the future to enhance the efficiency of this drug and check its impacts on humans in real-life scenarios. Further details of these trials will be made public in the future.